ASC Point Spread Function
From BioDIP
(Difference between revisions)
(→images) |
|||
| (14 intermediate revisions by one user not shown) | |||
| Line 1: | Line 1: | ||
| − | + | The point spread function (PSF) represents how an image of a point appears in a microscope. Measuring this PSF can reveal important informations: | |
| − | == | + | * angle of illumination axis |
| − | == | + | * damage/performance of objective |
| − | * | + | Having a system with a proper PSF is gets especially important for high resolution and deconvolution work. Analyzing the PSF of an objective can be semi-automated with the "PSF macro" by Laurent Gelman ([http://www.fmi.ch/ FMI Basel (Switzerland)]). |
| − | * | + | == requirements == |
| − | * the | + | * green fluorescent bead sample, preferably sub-resolution (170 nm) |
| − | == | + | == acquisition == |
| − | * | + | * use high resolution objectives (= the ones that people use for high resolution work) |
| − | + | * set up system for z-stack fluorescence | |
| − | + | ** blue excitation, green detection (i.e. EX 488 nm, EM 500-550 nm) | |
| − | + | ** pinhole at 1 airy unit | |
| − | * ( | + | ** pixel size of 100 nm |
| − | + | ** Z-step size of 200 nm | |
| − | * Fiji | + | ** 100 planes, focal plane of the bead in the middle |
| − | * | + | ** no oversaturated pixels |
| − | * | + | * scan field should be located in the center of the field of view (no panning, put the bead roughly in center by eye and stage) |
| − | === load bead stack | + | * size of the scan field should not be smaller than 20x20 µm, with the bead in the center (simplifies the analysis) |
| − | * | + | == prepare PSF macro == |
| − | * | + | * download the [[media:Gelman-psf-macro-v4.zip|PSF macro zip file]] and unzip it |
| − | * if necessary, split channels (Fiji > Image > Color > Split Channels) | + | * include the LUT: |
| − | * | + | ** Windows: locate the Fiji.app folder (i.e. C:\Program Files\Fiji.app), create a folder ''luts'' inside (if it's not there yet), place the ''LUTforPSFs.lut'' there |
| − | + | ** Mac: locate the Fiji.app folder (i.e. Applications\Fiji), right click on it > ''Show package content'', create a folder ''luts'' (if it's not there yet), place the ''LUTforPSFs.lut'' there | |
| − | + | * install the macro: | |
| − | + | ** open Fiji | |
| − | + | ** Plugins > Macros > Install... | |
| − | + | ** the macro appears in the Macros menu | |
| − | + | ** procedure has to be repeated for every new Fiji session | |
| − | + | == analysis == | |
| − | + | * load the bead stack into Fiji | |
| − | + | ** make sure the metadata is still there (i.e. that the image is scaled) | |
| − | + | * check the pixel size: Image > Show Info > Voxel size X | |
| − | + | * if necessary, crop image to a single bead (but also consider the next point) | |
| − | + | * the macro attempts to crop a 15x15 µm area with the bead in center; if the image dimensions are smaller, extend the area with black: Fiji > Image > Adjust > Canvas Size... | |
| − | + | * if necessary (multi-channel data), split channels (Fiji > Image > Color > Split Channels) | |
| − | + | * zoom into the image, in a way that you can hit the center of the bead w/o problems | |
| − | + | * start the macro: Plugins > Macros > gelman-psf-macro-v4 | |
| − | A. Selects the plane with the highest pixel intensity, adjusts display settings, | + | * enter the microscope information and click OK |
| − | opens the information dialog box. | + | * '''right'''-click into the center of the bead |
| − | + | ** that's the tricky step: the analysis will be wrong if you don't hit the center | |
| − | 1. Enter information in the dialog window which popped up. | + | ** depending on the system configuration you might need to first right-click into the center, and then left-click on the image window to get the process startet |
| − | + | * wait for the analysis to be done, result will be a stack of 2 images (see examples below) | |
| − | 2. Zoom in the image to clearly localize the center of the bead (you can also | + | == PSF macro overview: automatic macro actions / user actions == |
| − | navigate between planes if needed). | + | ''by Laurent Gelman'' |
| − | + | * A. Selects the plane with the highest pixel intensity, adjusts display settings, opens the information dialog box. | |
| − | 3. Right clicks with the mouse on the center of the bead. | + | * 1. Enter information in the dialog window which popped up. |
| − | + | * 2. Zoom in the image to clearly localize the center of the bead (you can also navigate between planes if needed). | |
| − | B. Crops the image to get 15mmx15mm area centered over the pixel clicked by the user. | + | * 3. Right clicks with the mouse on the center of the bead. |
| − | + | * B. Crops the image to get 15mmx15mm area centered over the pixel clicked by the user. | |
| − | C. Makes projections in X and Y of the stack | + | * C. Makes projections in X and Y of the stack |
| − | + | * D. Stitches together the cropped area and the projections | |
| − | D. Stitches together the cropped area and the projections | + | * E. Estimates and subtracts background |
| − | + | * F. Takes the square root of the image (to minimize photon noise and to mimic a decrease in histogram gain) | |
| − | E. Estimates and subtracts background | + | * G. Resizes the image to 550x550 pixels, adjusts display, changes LUT and displays the picture with a standardized name: ''Date_Scopename_Magnification_NA'' |
| − | + | ||
| − | F. Takes the square root of the image (to minimize photon noise and to mimic a | + | |
| − | decrease in histogram gain) | + | |
| − | + | ||
| − | G. Resizes the image to 550x550 pixels, adjusts display, changes LUT and | + | |
| − | picture | + | |
| − | + | ||
== images == | == images == | ||
<gallery> | <gallery> | ||
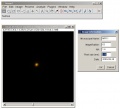
file:Set parameters.jpg|PSF macro: parameters dialog, open bead stack | file:Set parameters.jpg|PSF macro: parameters dialog, open bead stack | ||
| − | file:2009-09-29 MPI11 63x 1.4 FWHMa 3822nm - FWHMl 456nm.jpg|PSF macro result 1: 500 nm bead, confocal | + | file:2009-09-29 MPI11 63x 1.4 FWHMa 3822nm - FWHMl 456nm.jpg|PSF macro result 1: 500 nm bead*, confocal |
| − | file:2009-09-29 MPI11 63x 1.4 FWHMa 3822nm - FWHMl 456nm graphs.jpg|PSF macro result 2: 500 nm bead, confocal | + | file:2009-09-29 MPI11 63x 1.4 FWHMa 3822nm - FWHMl 456nm graphs.jpg|PSF macro result 2: 500 nm bead*, confocal |
</gallery> | </gallery> | ||
| + | (*) 500 nm bead = still above resolution limit! Use 170 nm beads to get a proper PSF. | ||
| + | [[category:ASC]] | ||
| + | __NOTOC__ | ||
Latest revision as of 14:03, 1 October 2009
The point spread function (PSF) represents how an image of a point appears in a microscope. Measuring this PSF can reveal important informations:
- angle of illumination axis
- damage/performance of objective
Having a system with a proper PSF is gets especially important for high resolution and deconvolution work. Analyzing the PSF of an objective can be semi-automated with the "PSF macro" by Laurent Gelman (FMI Basel (Switzerland)).
[edit] requirements
- green fluorescent bead sample, preferably sub-resolution (170 nm)
[edit] acquisition
- use high resolution objectives (= the ones that people use for high resolution work)
- set up system for z-stack fluorescence
- blue excitation, green detection (i.e. EX 488 nm, EM 500-550 nm)
- pinhole at 1 airy unit
- pixel size of 100 nm
- Z-step size of 200 nm
- 100 planes, focal plane of the bead in the middle
- no oversaturated pixels
- scan field should be located in the center of the field of view (no panning, put the bead roughly in center by eye and stage)
- size of the scan field should not be smaller than 20x20 µm, with the bead in the center (simplifies the analysis)
[edit] prepare PSF macro
- download the PSF macro zip file and unzip it
- include the LUT:
- Windows: locate the Fiji.app folder (i.e. C:\Program Files\Fiji.app), create a folder luts inside (if it's not there yet), place the LUTforPSFs.lut there
- Mac: locate the Fiji.app folder (i.e. Applications\Fiji), right click on it > Show package content, create a folder luts (if it's not there yet), place the LUTforPSFs.lut there
- install the macro:
- open Fiji
- Plugins > Macros > Install...
- the macro appears in the Macros menu
- procedure has to be repeated for every new Fiji session
[edit] analysis
- load the bead stack into Fiji
- make sure the metadata is still there (i.e. that the image is scaled)
- check the pixel size: Image > Show Info > Voxel size X
- if necessary, crop image to a single bead (but also consider the next point)
- the macro attempts to crop a 15x15 µm area with the bead in center; if the image dimensions are smaller, extend the area with black: Fiji > Image > Adjust > Canvas Size...
- if necessary (multi-channel data), split channels (Fiji > Image > Color > Split Channels)
- zoom into the image, in a way that you can hit the center of the bead w/o problems
- start the macro: Plugins > Macros > gelman-psf-macro-v4
- enter the microscope information and click OK
- right-click into the center of the bead
- that's the tricky step: the analysis will be wrong if you don't hit the center
- depending on the system configuration you might need to first right-click into the center, and then left-click on the image window to get the process startet
- wait for the analysis to be done, result will be a stack of 2 images (see examples below)
[edit] PSF macro overview: automatic macro actions / user actions
by Laurent Gelman
- A. Selects the plane with the highest pixel intensity, adjusts display settings, opens the information dialog box.
- 1. Enter information in the dialog window which popped up.
- 2. Zoom in the image to clearly localize the center of the bead (you can also navigate between planes if needed).
- 3. Right clicks with the mouse on the center of the bead.
- B. Crops the image to get 15mmx15mm area centered over the pixel clicked by the user.
- C. Makes projections in X and Y of the stack
- D. Stitches together the cropped area and the projections
- E. Estimates and subtracts background
- F. Takes the square root of the image (to minimize photon noise and to mimic a decrease in histogram gain)
- G. Resizes the image to 550x550 pixels, adjusts display, changes LUT and displays the picture with a standardized name: Date_Scopename_Magnification_NA
[edit] images
(*) 500 nm bead = still above resolution limit! Use 170 nm beads to get a proper PSF.