Physical exercise increases Notch activity, proliferation and cell cycle exit of type-3 progenitor cells in adult hippocampal neurogenesis.
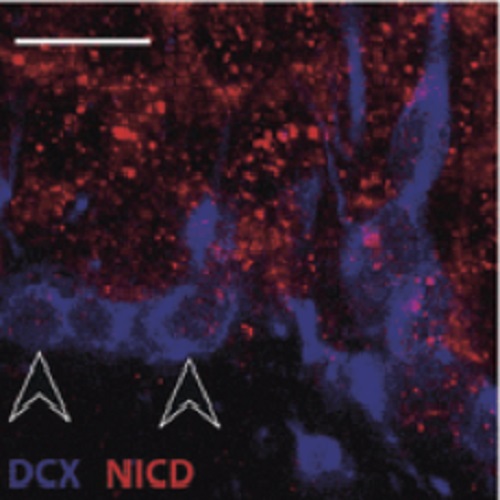
In adult hippocampal neurogenesis of mice, the proliferation of precursor cells can be stimulated by voluntary exercise (wheel-running). Physical activity has an additional effect on late progenitor cells (type-3) by promoting cell survival and further maturation. Notch1 is a key regulator of various steps in neuronal development, including the inhibition of cell cycle exit and neuronal differentiation of neural stem cells, as well as promoting the survival and dendritic branching of newborn neurons. We here report that physical activity increased the proportion and absolute number of doublecortin(+) (DCX) type-2b and type-3 progenitor cells that showed an activated Notch1 pathway. In contrast, the fraction of dividing cells with nuclear Notch intracellular domain expression indicating an activated Notch pathway was not affected by physical exercise. We used double labeling with two halogenated thymidine analogs, iododeoxyuridine and chlorodeoxyuridine, to distinguish between cell cycle exit and continued division at the progenitor cell level. After 7 days of physical exercise, the proliferative activity of precursor cells was increased, whereas the proportion of type-2b/3 cells re-entering S-phase was reduced. Consistent with this observation, the proportion of DCX(+) cells that expressed the marker of postmitotic immature granule cells (calretinin) was enhanced. Running promotes both the proliferation and cell cycle exit of DCX(+) type-3 precursors, possibly by preferentially stimulating a last neurogenic cell division. These pro-proliferative effects are independent of Notch1, whereas the running-induced survival and cell cycle exit of type-3 progenitor cells might by mediated by Notch1 activity.
- Eur. J. Neurosci. 2010 Oct 08;32(8):1256-64
- 2010
- Medical Biology
- 20950279
- PubMed
Enabled by:
