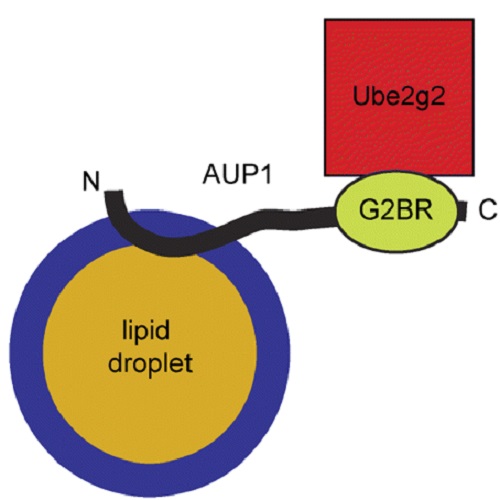
Ancient ubiquitous protein 1 (AUP1) localizes to lipid droplets and binds the E2 ubiquitin conjugase G2 (Ube2g2) via its G2 binding region.
Lipid droplets (LDs), the major intracellular storage sites for neutral lipids, consist of a neutral lipid core surrounded by a phospholipid monolayer membrane. In addition to their function in lipid storage, LDs participate in lipid biosynthesis and recently were implicated in proteasomal protein degradation and autophagy. To identify components of the protein degradation machinery on LDs, we studied several candidates identified in previous LD proteome analyses. Here, we demonstrate that the highly conserved and broadly expressed ancient ubiquitous protein 1 (AUP1) localizes to LDs, where it integrates into the LD surface in a monotopic fashion with both termini facing the cytosol. AUP1 contains a C-terminal domain with strong homology to a domain known as G2BR, which binds E2 ubiquitin conjugases. We show that AUP1, by means of its G2BR domain, binds to Ube2g2. This binding is abolished by deletion or mutation of the G2BR domain, although the LD localization of AUP1 is not affected. The presence of the AUP1-Ube2g2 complex at LDs provides a direct molecular link between LDs and the cellular ubiquitination machinery.
- J. Biol. Chem. 2011 Feb 18;286(7):5599-606
- 2011
- Cell Biology
- 21127063
- PubMed
Enabled by:
