PHD4 stimulates tumor angiogenesis in osteosarcoma cells via TGF-α.
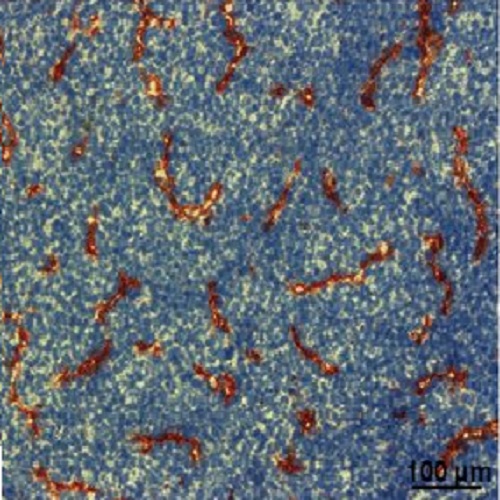
Solid tumor growth is intimately associated with angiogenesis, a process that is efficiently triggered by hypoxia. Therefore, oxygen-sensitive signaling pathways are thought to play a critical role in tumor angiogenesis and progression. Here, the function of prolyl hydroxylase-4 (PHD4), a relative of the prolyl hydroxylase domain proteins 1-3 that promote the degradation of hypoxia-inducible factors (HIF), was interrogated. To test the hypothesis that PHD4 might inhibit tumor angiogenesis, it was overexpressed in osteosarcoma cells, and unexpectedly, this manipulation led to increased tumor blood vessel density. However, the newly formed blood vessels were smaller than normal and appeared to be partially nonfunctional, as indicated by poor vessel perfusion. PHD4 overexpression in tumor cells stimulated the expression of TGF-α, which was necessary and sufficient to promote angiogenic sprouting of endothelial cells. On the other hand, PHD4 overexpression reduced HIF-2α protein levels, which in turn inhibited in vivo tumor growth. Combined, elevated PHD4 levels deregulate angiogenesis via increased TGF-α expression in vitro and in vivo. These data support the hypothesis that tumor growth can be uncoupled from vessel density and that the individual PHD family members exert distinct functions in tumors. IMPLICATIONS: PHD4 influences tumor growth and vascularization through discrete mechanisms and molecular pathways that likely have therapeutic potential.
- Mol. Cancer Res. 2013 Nov 18;11(11):1337-48
- 2013
- Medical Biology
- 24048703
- PubMed
Enabled by:
